Rye is a species of grass related to wheat and barley. Yet, within their unassuming yellow grains, they hold a valuable secret that’s taken three decades to decipher: mysteriously, it could stop cancerous tumour growth, but no one knew how. A team of researchers at Northwestern University, led by Karl A. Scheidt, embarked on a quest to unearth those secrets. Yet, to tell their story, we must first return to the past.
Molecular Mystery
1997. University of Geneva, Switzerland. A group of researchers was examining a medicine derived from rye pollen known as Cernilton, an effective treatment for a non-cancerous enlargement of the prostate. They were searching for precisely what within Cernilton could reduce tumors. To their amazement, they discovered a set of molecules that could inhibit tumour growth, including cancerous tumour growth, within living creatures. They named these molecules “secalosides”, a combination of the Latin name of rye, secale cerale, and the type of molecule it was, a glycoside. Unfortunately, their creativity ended there, as the different secalosides discovered were subsequently labelled A, B, C, and D. Logically, the next step would be to determine how the secalosides restrained cancer cell growth, and to do so, the scientists needed to figure out what the secalosides looked like.
This is where they hit a roadblock.
These secolasides came in pairs, two sets of twins. On first glance, each twin may seem identical to one another, A the spitting image of B, and C the spitting image of D. Upon closer examination, the secalosides were actually a pair of compounds known as “epimers”: almost identical, save for the fact that one part of them was flipped. One twin had a mole on her left cheek, and the other had a mole on her right. Trouble arose when the scientists attempted to determine the structure of Secolasides A/B:
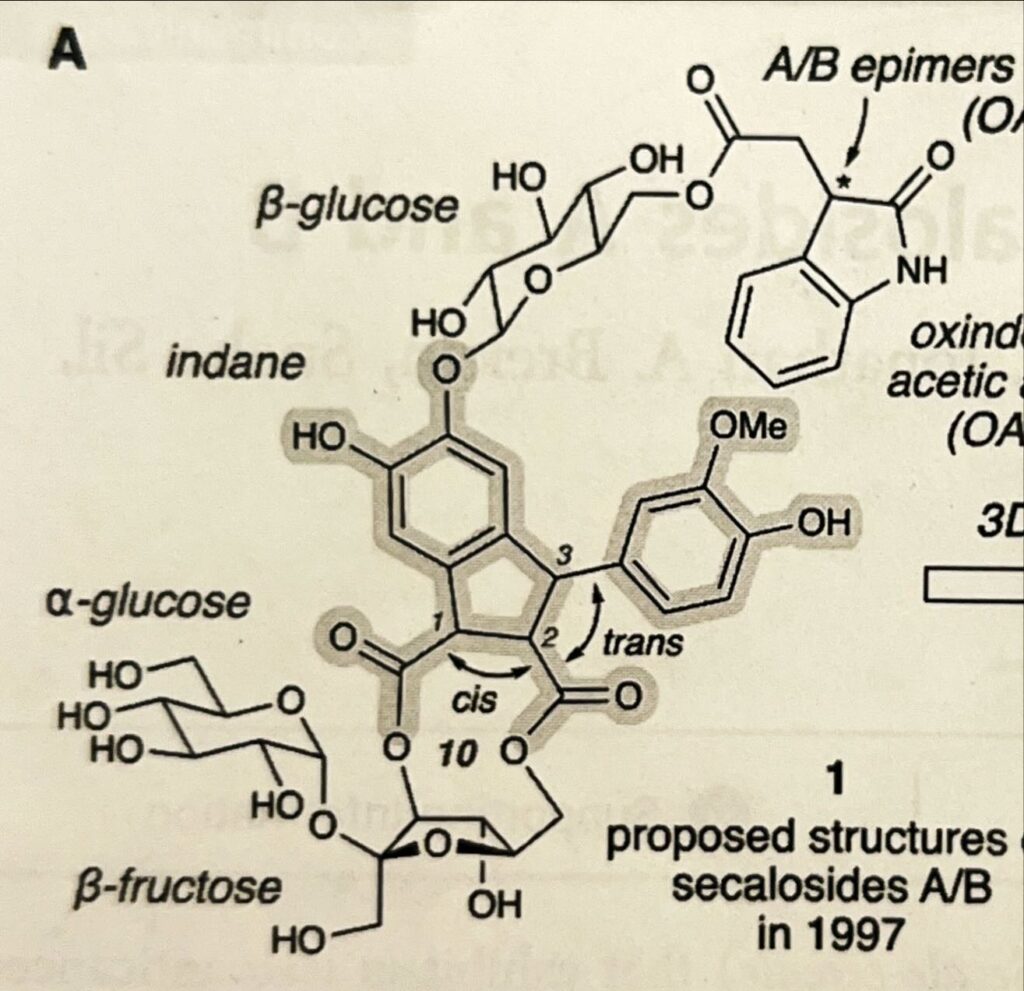
(J. Am. Chem. Soc. 2026, 148, 86-92)
What do you see? Hexagons, pentagons, letters all over the place. To the imaginative, it may even look like the face of a Minecraft creeper or a particularly upset pug. Although they’d identified its atoms and groups, and the 2-dimensional layout of the molecule, secalosides A/B remained as elusive as ever when it came to its 3-dimensional structure. It was as if a group of archeologists had unearthed a new and entirely complete fossil, pieced together where each bone went, but were at a loss when it came to assembling it in three dimensions. Did its arm go up, or go down? How did the thigh bone connect to the hip? No one knew. Biological interactions depend heavily on the shape. Frustratingly, as the true form of this tiny, cancer-inhibiting, molecular dinosaur remained unknown, our understanding of its mysterious anticancer abilities slipped out of reach. Further research ground to a halt. That is, of course, until 2025.
Rings and Riddles
Fast forward thirty years, Karl A. Scheidt and co-workers decide to embark on the challenge to unveil the secalosides’ true structure. Techniques usually used to determine molecular structure, such as nuclear magnetic resonance, weren’t enough. Therefore, the researchers decided to simply remake them from scratch: piecing together secalosides A/B brick by brick would certainly reveal what they looked like. To start, Karl A. Scheidt and co-workers settled on two potential 3D structures for the secalosides and planned to test which one of the two behaved identically to an extracted sample of the molecule, therefore identifying its true form. Although they’d significantly narrowed down their options, the tricky step would be synthesizing the molecule itself.
Two challenges immediately presented themselves: in order to keep track of the changes made to the molecule’s 3D structure, the chemists would need remarkable control over how they physically pieced together the molecule. It’s simple when you’re putting blocks of LEGO where they’re supposed to go, but much, much harder when the thing you’re building is 50,000 times smaller than the width of a single hair. Secondly, the secalosides hid their true form underneath a veil of incredible structural complexity. Perhaps, if you’ve studied organic chemistry before, you may see the peculiar 10-membered ring, right below the pug’s mouth at the bottom of the molecule. Conveniently, it is labelled “10”. Atoms, particularly carbon, love to form rings. Happy, stable 6-membered or 5-membered rings are the most common. Anything smaller and the carbons are forced into strained, uncomfortable positions, and anything larger results in a ring too flimsy to stay together. Not only was this a naturally occurring 10-membered ring, but it was one with many other groups sticking out of it, increasing the structural strain. Any attempts to directly form the ring from smaller building blocks, such as wrapping a long chain of carbons into a circle and knotting it like a bracelet, fell apart.
Switching gears, the scientists decided to fight fire with fire: they would start with a larger ring instead of smaller building blocks, and then shrink the ring to the desired size. They chose the 13-membered ring because it was both more stable due to its flexibility and because once they shrank it down, it would form both the ten-membered ring and the five-membered one above it, hitting two birds with one stone.
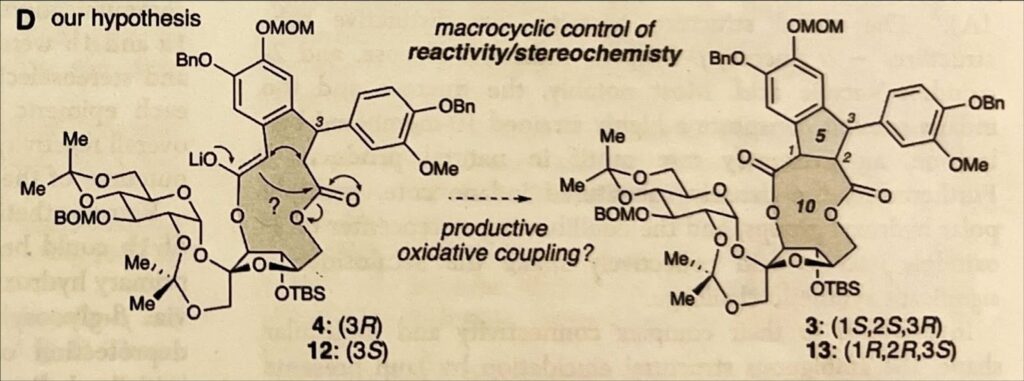
(J. Am. Chem. Soc. 2026, 148, 86-92)
Yet, there was a risk: the large ring wouldn’t shrink to the 10-membered ring, but instead go straight towards a far more stable ring, such as a six-membered ring, rather than the desired 10-membered one, known as Diekmann condensation. When the chemists tested this using smaller, acyclic (not rings) molecules as models, Dieckmann condensation was precisely what happened.
In spite of this, the ring contraction felt too clean to be completely abandoned. The chemists theorized that maybe, just maybe, the bulky ring would behave differently compared to its smaller, non-cyclic counterparts. Unlike a regular chain, the ring had limited wriggle room and would have to fold itself in a specific way to prevent all the groups sticking out of it from clashing into one another. Potentially, this position would block Dieckmann condensation, forcing the 10-membered ring to form. If their hypothesis was right, the 13-membered ring also served as a scaffold, giving them structural control over where things went.
They were correct! The 13-membered ring shrank in exactly the way the chemists hypothesized it would, leaving not even a speck of undesired product! With that, they had a 10-membered ring in their hands, and after a few more steps, they formed what they believed could be the elusive secalosides. Was this it? Did the chemists not only find but also make the mysterious secalosides, thirty decades after they were discovered?
Everybody held their breath, the results of the spectral data — a molecule’s “fingerprint” — came out as… inconclusive. The molecule that they’d created was not, in fact, secalosides A/B.
Back to the drawing board.
Except, not quite. They’d already crossed the largest barrier of synthesizing the secalosdies: that pesky 10-membered ring. As they’d narrowed down the potential secalosides to two molecules, all that was left to do was to shuffle some groups around. Although that in of itself presented some challenges, they soon procured the second molecule, which had spectral data that perfectly matched that of secalosides A/B!
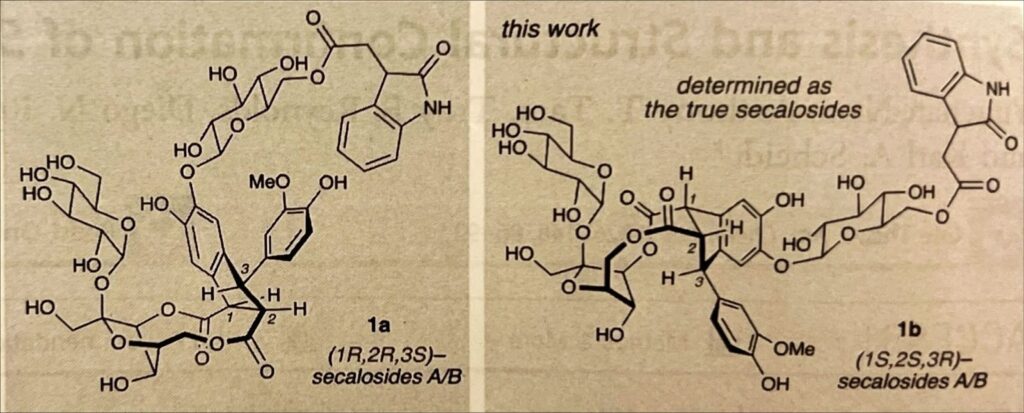
(J. Am. Chem. Soc. 2026, 148, 86-92)
Rye is this important?
Scheidt and team discovered both the molecular structure of the secalosides A/B and the steps taken to create them at once! They embarked on a journey to create an incredibly complex molecule, but also one that they didn’t even know the real structure of. No matter what challenges they faced, they pushed forward, coming up with creative and innovative approaches to overcome whatever roadblocks came their way. Yet, aside from being an impressive feat, the synthesis and structural determination of secalosides A/B has finally revealed the secret in the rye, re-opening the investigation on the secalosides’ cancer-inhibiting abilities! We no longer have to ask ourselves what secalosides are, but instead can finally move on to discovering how they interact with cancerous cells, and how we can harness their power to save lives.
Sources:
Jaton, J.-C., Roulin, K., Rose, K., Sirotnak, F. M., Lewenstein, A., Brunner, G., Fankhauser, C. P.,
& Burger, U. (1997). The secalosides, novel tumor cell growth inhibitory glycosides from a
pollen extract. Journal of Natural Products, 60(4), 356-360. https://doi.org/10.1021/np9606557
Nam, Y., Tam, A. T., Reynolds, T. E., Rojas, D. N., Brekan, J. A., Sil, S., & Scheidt, K. A. (2025).
Synthesis and structural confirmation of secalosides A and B. Journal of the American ChemicalSociety, 148(1), 86-92. https://doi.org/10.1021/jacs.5c1886z